Ophthalmology
Voiant is the industry leader in providing the highest quality data for ophthalmology and ocular safety trials.
Since 1998, the Digital Angiography Reading Center (DARC) has been the world leader in retinal imaging evaluation services, supporting over 200 global studies. Now part of Voiant, and strengthened by the recent acquisition of Voxeleron, we unite over 30 years of clinical excellence with state-of-the-art AI-driven image analysis. This powerful combination enhances our ability to deliver precise, efficient imaging solutions —accelerating the development of innovative treatments and improving outcomes for patients worldwide.
VOIANT PRESCREEN™
Voiant’s new AI-image analysis feature enables sites to determine potential patient eligibility in real-time, reducing screen failure rates in ophthalmic studies and the time between screening and randomization, ultimately saving thousands of dollars in unnecessary screening procedures and accelerating study timelines.
VOIANT TREATMENT DETERMINATION™
Voiant’s new AI-image analysis feature enables principal investigators (PI) to obtain real-time precision measurements of imaging studies to determine whether protocol-specific criteria for retreatment or rescue are met, empowering the PI with the tools they need to make timely, informed decisions for their patients.
AI-Powered Excellence in
Ophthalmology Imaging Technology
We combine advanced imaging technologies with AI-driven analysis to accelerate biomarker discovery in ophthalmology. By integrating imaging modalities with biomolecular insights, we identify novel indicators of disease progression, treatment response, and patient-specific outcomes. Our AI capabilities refine and validate these biomarkers, unlocking the full potential of ophthalmic imaging to drive smarter, faster clinical trials. This includes advanced techniques such as:
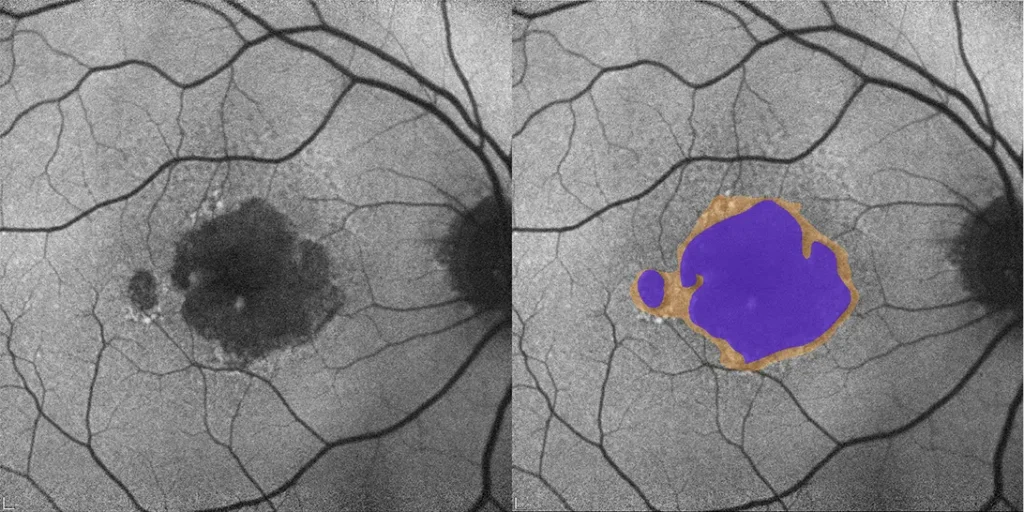
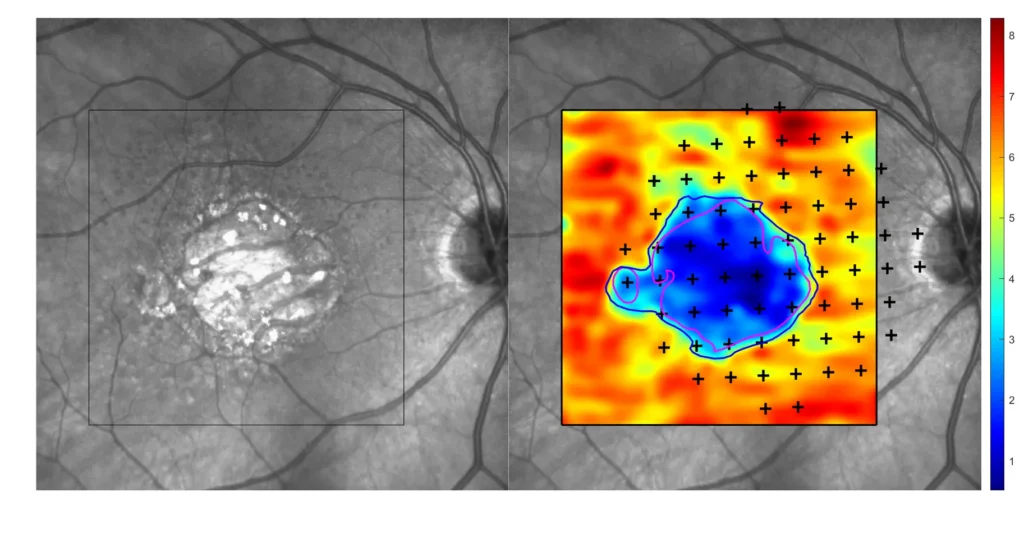
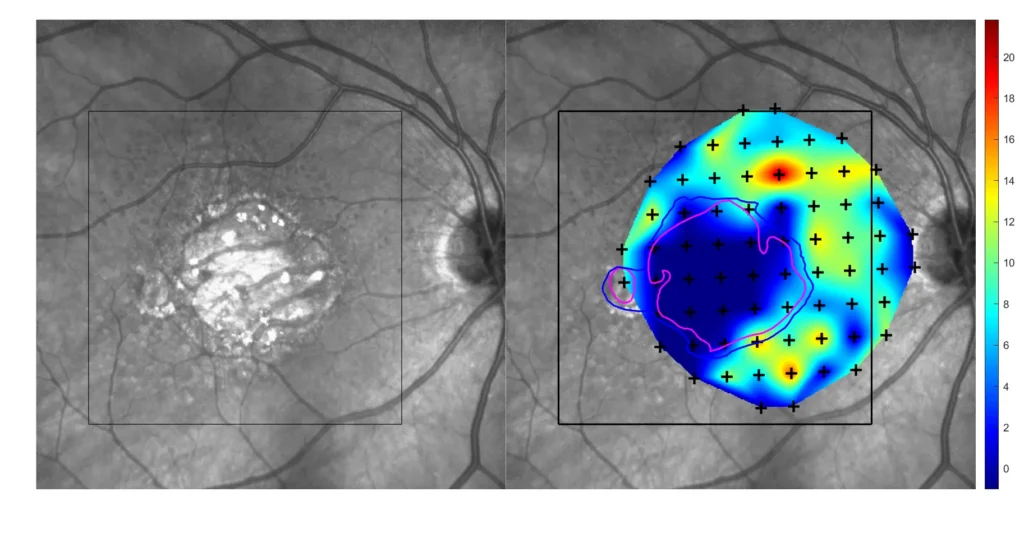
Our multimodal overlays combine structural and functional imaging modalities, such as OCT, FAF, and microperimetry, to provide a more comprehensive understanding supported by expert-trained readers who interpret across modalities with deep disease insight.
Ophthalmology Imaging Expertise
Our work spans a broad range of ophthalmic indications, imaging modalities, and an expanding portfolio of biomarkers. From acquisition to analysis, we integrate advanced imaging technologies and tailored data outputs to support precise, efficient, and impactful trial outcomes.
Experience Across Ophthalmology Indications
Voiant has delivered imaging services on Phase I-IV drug and medical device trials across many ophthalmic indications, including:
Posterior Segment Disorders
- Age-related Macular Degeneration (AMD)
- Intermediate
- Advanced – Neovascular and Geographic Atrophy (GA)
- Diabetic Retinopathy
- Non-Proliferative
- Proliferative
- Retinal Vascular Occlusion
- Macular Edema
- Diabetic
- Non-Diabetic Inherited Retinal Diseases including:
- Retinitis Pigmentosa
- Stargardt Disease
- Ocular Melanoma
- Optic Nerve Disorders including:
- Glaucoma
- Optic Neuropathy
- Uveitis
Anterior Segment Disorders
- Cataract
- Eyelid Margin Disease including:
- Blepharitis
- Meibomian Gland Dysfunction (MGD)
- Ocular Surface Disease including:
- Conjunctivitis
- Dry Eyes
- Ulceration
Complimentary Modality Services
Voiant has extensive expertise across ocular imaging modalities designed to meet the evolving needs of trials, including:
- Optical Coherence Tomography (OCT):
- Posterior Pole – Macula, RNFL, ONH
- Anterior Segment (AS-OCT)
- Enhanced Depth Imaging (EDI-OCT)
- Spectral Domain (SD-OCT)
- Swept-Source (SS-OCT)
- Widefield (WF-OCT) / Area of Interest (AOI)
- Angiography:
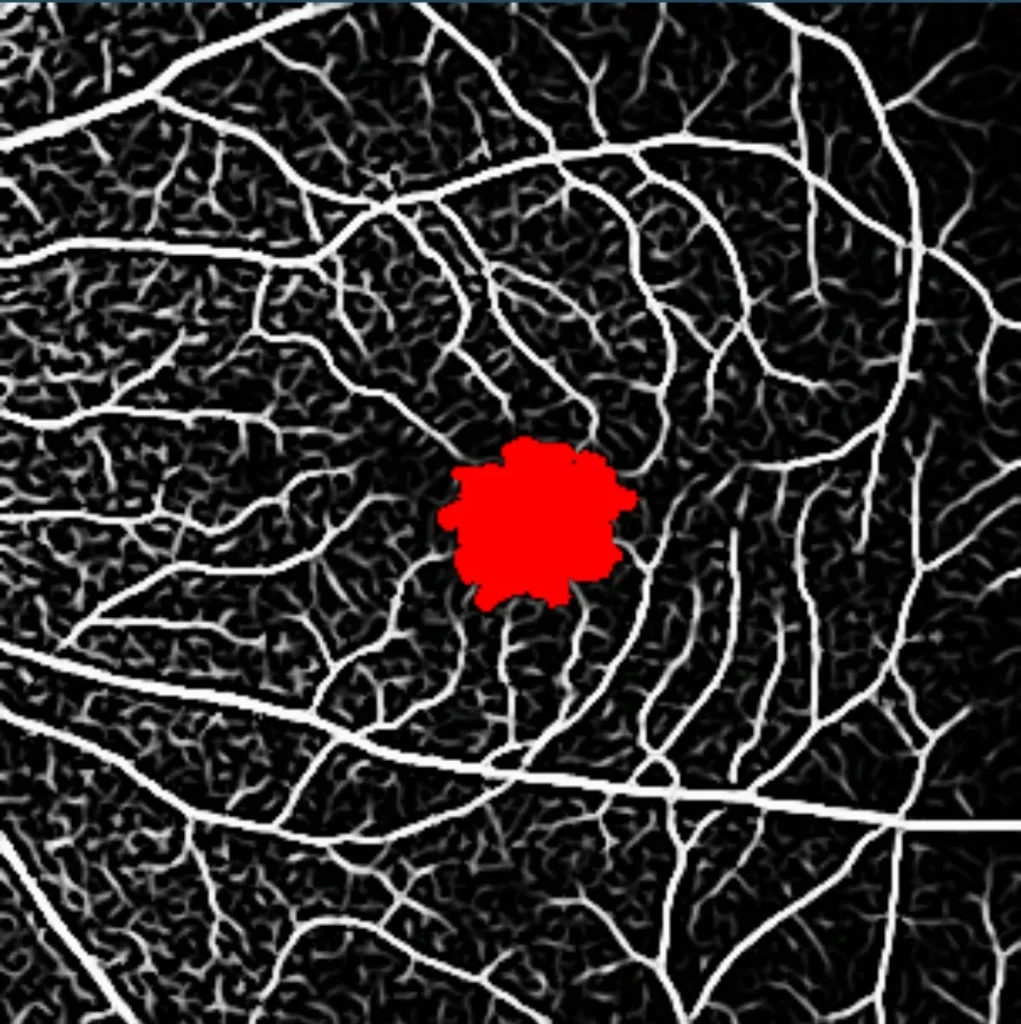
- OCT – Angiography (OCT-A)
- Fluorescein Angiography (FA)
- Indocyanine Green (ICG) Angiography
- B-Scan Ultrasonography
- Color Fundus Photography (CFP)
- Electroretinography (ERG):
- Full Field (ffERG)
- Multifocal (mfERG)
- Pattern (pERG)
- Photopic Negative Response (phNR)
- Visual Evoked Potential (VEP)
- Flavoprotein Fluorescence (FPF)
- Fundus Autofluorescence (FAF)
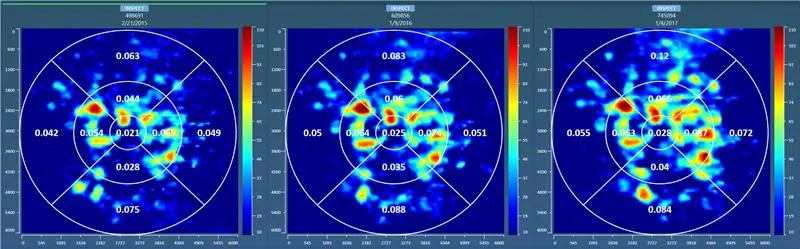
- Microperimetry (MP)
- Visual Field Perimetry (VFP) – 10-2, 24-2, 30-2
- Corneal Confocal Microscopy (CCM)
- Specular Microscopy (SM)
- Near Infrared Imaging (IR)
- Slit Lamp Photography (SLP) – Conjunctiva, Cornea, Iris, Lens
Expanding Biomarker Capabilities
Voiant has built a robust and growing set of biomarkers to enhance trial outcomes, including:
- Centration Correction
- Segmentation Correction of 8 Standard Retinal Layers + Optional Customization
- Customized Macular Volume Assessments
- Customized Central Subfield Thickness Assessments
- Umbo Values Measurements
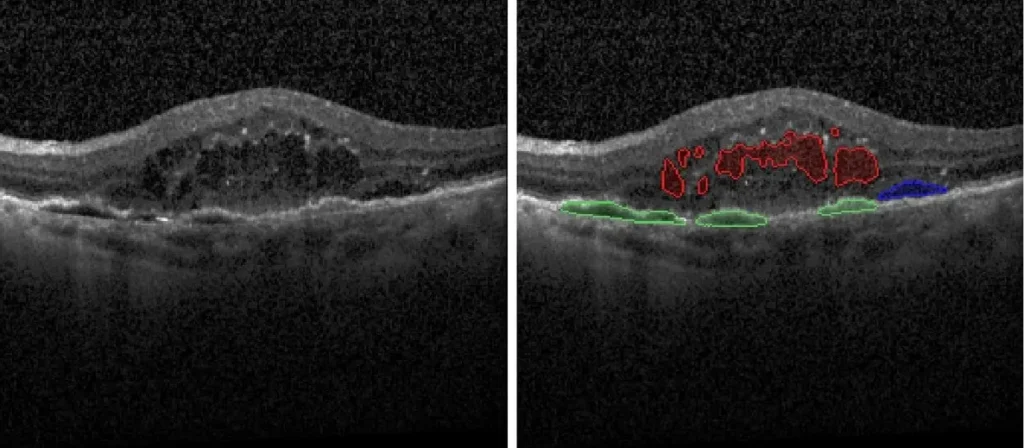
- Geographic Atrophy (GA) Segmentation and Quantification
- Ellipsoid Zone (EZ) Thickness and Loss Area
- En Face Measurements
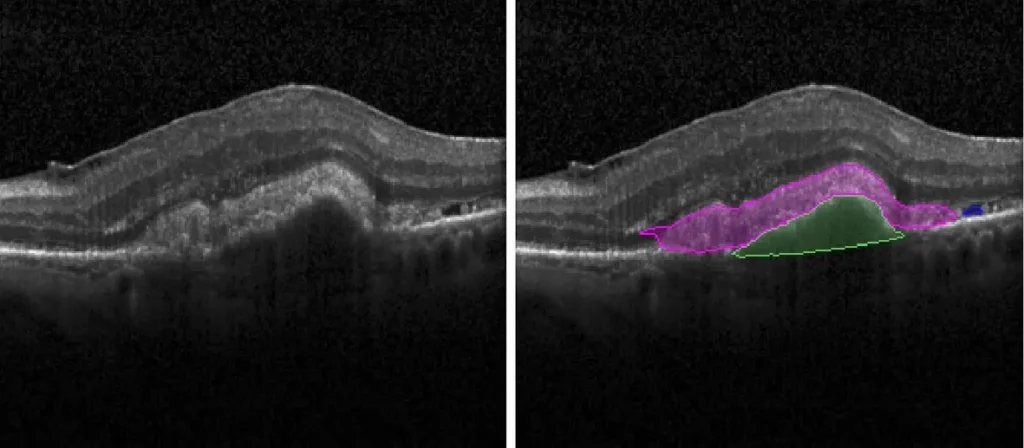
- Fluid Segmentation with Linear, Area, and Volume Quantification:
- Intraretinal Fluid (IRF)
- Subretinal Fluid (SRF)
- Pigment Epithelial Detachment (PED)
- Subretinal Hyper-Reflective Material (SHRM)
- Drusen Quantification
- Hyper-Transmission Defect Detection/Quantification
- Progression Tracking
- Multimodal Overlay
- Automated Image Scaling
- Automated Assessment/Measurement of Pathology
- Disorganization of Retinal Inner Layers (DRIL) Assessment
- Quantification of Fundus Hyper-Autofluorescence
- Fluid Dynamics
- Constructive and Actionable Site Quality/Certification Feedback
- Customizable Biomarkers
Ocular Safety Study Package
Alongside ophthalmic drug trials, we’re highly experienced in evaluating ocular toxicities from systemic drugs in oncology, CNS, and other areas. Our deep knowledge of eye health led to the creation of a robust Ocular Safety Study Package, helping sponsors confidently run safety studies with reliable results. Supported by our white-glove service, our package includes:
- Comprehensive, hands-on support for sites new to ophthalmic imaging through every step of the process
- Protocol consultation with highly qualified medical experts for ocular safety assessment
- Accelerated site certification for ophthalmologic image collection
- Rapid analysis and safety reports
- Guidance and support with regulatory agencies
Examples of indications we’ve supported include:
- General Safety
- Anti-Malarial
- Advanced Solid Tumor
- Cholangiocarcinoma
- Hepatic Impairment
- Hepatitis B
- Major Depressive Disorder/Bipolar Disorder
- Metastatic Melanoma
- Multiple Sclerosis
- Nephrology
- Parkinson’s Disease
- Renal Impairment
- Urinary/Urothelial/Bladder
Your imaging partner from start to endpoint
Learn more about how Voiant’s innovative solutions and expert services can provide custom-tailored support for your next trial.